The C-cl Bond Dissociation Energy in Cf3cl
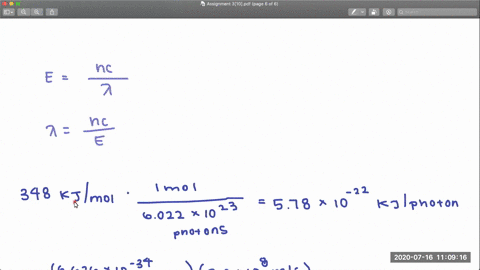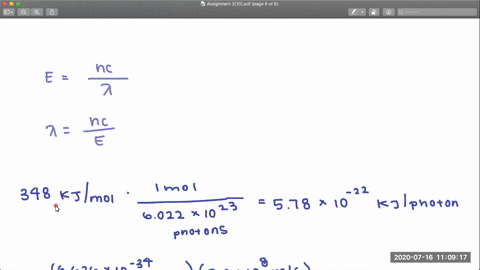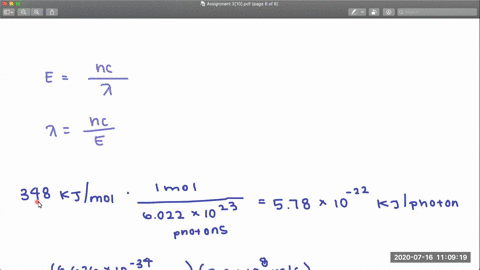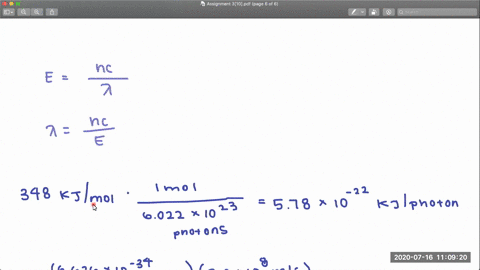
353 nm 248 nm. In CF 3 Cl the CCl bond-dissociation energy is 339 kJmol.

What Is The Difference Of The Bond Length Of C Cl Between Ch3 Cl And Cf3 Cl Quora
In CCl 4 the CCl bond-dissociation energy is 293 kJmol.

. CCl and CF bonds while the branching ratio elicited an energy-dependent relationship. 137 nmPlease explain indetails This problem has been solved. Of particular importance are the energies necessary to induce homolytic cleavage of the CCl bonds of such species ie the homolytic bond dissociation energies BDEs which culminates in the formation of carbon.
Profile fits require the fast subsequent dissociation of CFClâ and therefore the short-time kinetics can be best explained as being due to C-Cl bond breaking in the parent CFClâ. The CCl and CF bond dissociation energies in CF3Cl are 339 kJmol and 482 kJmol respectively. Please include units with your calculation.
Rotational centrifugal distortion and nuclear quadrupole 35 Cl and 37 Cl coupling constants have been precisely obtained from high-resolution measurements. What is the range of wavelengths of photons that can cause C-Cl bond rupture in one molecule but not in the other. The gas-phase equilibria Cl 2 CF 3 BrBrClCF 3 Cl 3 Br 2 CF 3 ClBrClCF 3 Br 4 Cl 2 C 2 F 5 BrBrClC 2 F 5 Cl 5 have been studied over the temperature ranges.
The CO bond dissociation energy in CO2 is 799 kJmol. Department of Energys Office of Scientific and Technical Information Ab Initio Calculations and Three Different Applications of Unimolecular Rate Theory for the Dissociations of CCl4 CFCl3 CF2Cl2 and CF3Cl Journal Article OSTIGOV. This in turn means that in H X 3 C C l the C C l bond will have more carbon-p character as an idealised sp³ orbital and is.
Information on the internal dynamics and the dissociation energy of the complex is provided. The homolytic M-Cl bond dissociation energy Dhomo ΔZPE is 817 1056 962 and 936 kcalmol for CH3-Cl SiH3-Cl GeH3-Cl and SnH3-Cl respectively NL-SCFTZ2P. The max wavelength of radiation required to rupture these bonds are ________ and _____ respectively.
Asked Jul 29 2018 in Chemistry by bonilla411. What is the maximumwavelength of photons that can rupture this bondA. Up to 256 cash back 28 Mar 2020.
The two subunits of the complex are held together via a ClC halogen bond interaction. Vertical excitation energies DeltaEvertical dissociation energies DeltaEdiss dissociation enthalpies DeltaHdiss and the oscillator strength f have also been computed. The effects of the Cl equatorial belt present in perhalogenated molecules such as CF3Cl have been hitherto overlooked in describing the origin of noncovalent interactions.
A comparison of bond dissociation energies of the type DCF 3. Besides its typical halogen donor behavior exhibiting a Cl σ-hole in forming ClB halogen bonds B is an electron-rich region CF3Cl reveals a new interaction site in its complex with CO2 when. What is the range of wavelengths of photons that can cause CCl bond.
Chlorinated organic molecules are ubiquitous and as such knowledge of their fundamental thermodynamic properties is desirable. The coincident time-of-flight mass spectra and three-dimensional time-sliced images of the CF 2 Cl fragment were recorded at a few specific photon energies. In CF3Cl the C-Cl bond-dissociation energy is 339 kJ mol.
Δ H f C g 121 k J m o l Δ H f C l g 716 k J m o l Δ H f C C l X 4 g 10648 k J m o l. What is the maximum wavelength of photons that can rupture this bond. The C-Cl and C-F bond dissociation energies in CF3Cl arw 339 kjmol and 482 kjmol respectively.
If you compare H X 3 C C l and F X 3 C C l then the C H bond has less carbon-p character than an idealised sp³ orbital while the C F bond will have more carbon-p character than the idealised sp³ orbital. PDF The dissociative photoionization of CF3Cl was investigated using threshold photoelectron photoion coincidence TPEPICO imaging in the energy. The c-cl bond dissociation energy in cf3cl is 339 kjmol.
A detailed analysis of the. The dissociative photoionization of CF 3 Cl was investigated using threshold photoelectron photoion coincidence TPEPICO imaging in the energy range of 12301850 eV. In CCl4 the bond-dissociation energy is 293 kJ mol.
Basically I already answered this but I was a bit confused about the result cause I dont know if there is a bond that is negative cause am familiar with the energy given to dissociation of a bond or the formation of it. The CF 3 fragment was dominant in the dissociation of the X 2E A A0 and B2A00 states while CF 2Cl. The CCl bonddissociation energy in CF3Cl is 339 kJmol.
Full geometry optimizations have been carried out for S0 as well as relaxed potential energy calculations for both states along the C-Cl bond distance. The CCl and CF bond dissociation energies in CF3Cl are 339 kJmol and 482 kJmol respectively. What is the maximum wavelength of photons that can cause C-Cl bond dissociation.
Additionally SET is known to be concerted with dissociation of the C-Cl bond in hydrogenolysis of many CACs 13 5758 and these can be described by. These results are compared with other published values of DCF 3 Cl and ΔH f CF 3 Cl. The maximum wavelengths of electromagnetic radiation required to rupture these bonds are ________ and ________ respectively.
The maximum wavelengths of electromagnetic radiation required to rupture these bonds are ________ and ________ respectively. 450 nm 207 nm. In CF3Cl the C-CI bond-dissociation energy is 339 KJmol.
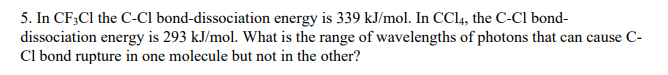
Solved 5 In Cf3cl The C Cl Bond Dissociation Energy Is 339 Chegg Com

A F Psd Spectrum Near The F1s Edge From Solid Cf 3 Cl B Total Download Scientific Diagram

9 3d Plots For Halogen Bonded Complexes A Cf 3 Cl Dme B Cf 3 Cl Dms

The Cl Loss Potential Energy Curves Of The Low Lying Electronic States Download Scientific Diagram

Tpepico Time Of Flight Mass Spectra Of The Dissociative Photoionization Of Cf 3 Cl At Various Photon Energies Where The Threshold Photoelectron Spectrum Is Shown In The Inserted Panel
Solved In Mathrm Cf 3 Mathrm Cl The Mathrm C Mathrm Cl Bond Dissociation Energy Is 339 Mathrm Kj Mathrm Mol In Mathrm Ccl 4 The Mathrm C Mathrm Cl Bond Dissociation Energy Is 293 Mathrm Kj Mathrm Mol What Is The Range

The Cl Loss Potential Energy Curves Of The Low Lying Electronic States Download Scientific Diagram

Solved 50 5pts What Is The Ph Of A Solution That Contains Chegg Com

Comparison Of Bond Dissociation Energies A T 298 K Derived From Download Table

The Cl Loss Potential Energy Curves Of The Low Lying Electronic States Download Scientific Diagram

F Loss Potential Energy Curves Of The Low Lying Electronic States Of Cf Download Scientific Diagram

Dissociative Photoionization Of Cf 3 Cl Via The C 2 E And D 2 E States Competition Of The C F And C Cl Bond Cleavages Physical Chemistry Chemical Physics Rsc Publishing Doi 10 1039 C8cp07332e
Answered In Cf3cl The C Cl Bond Di S Sociat Ion Bartleby
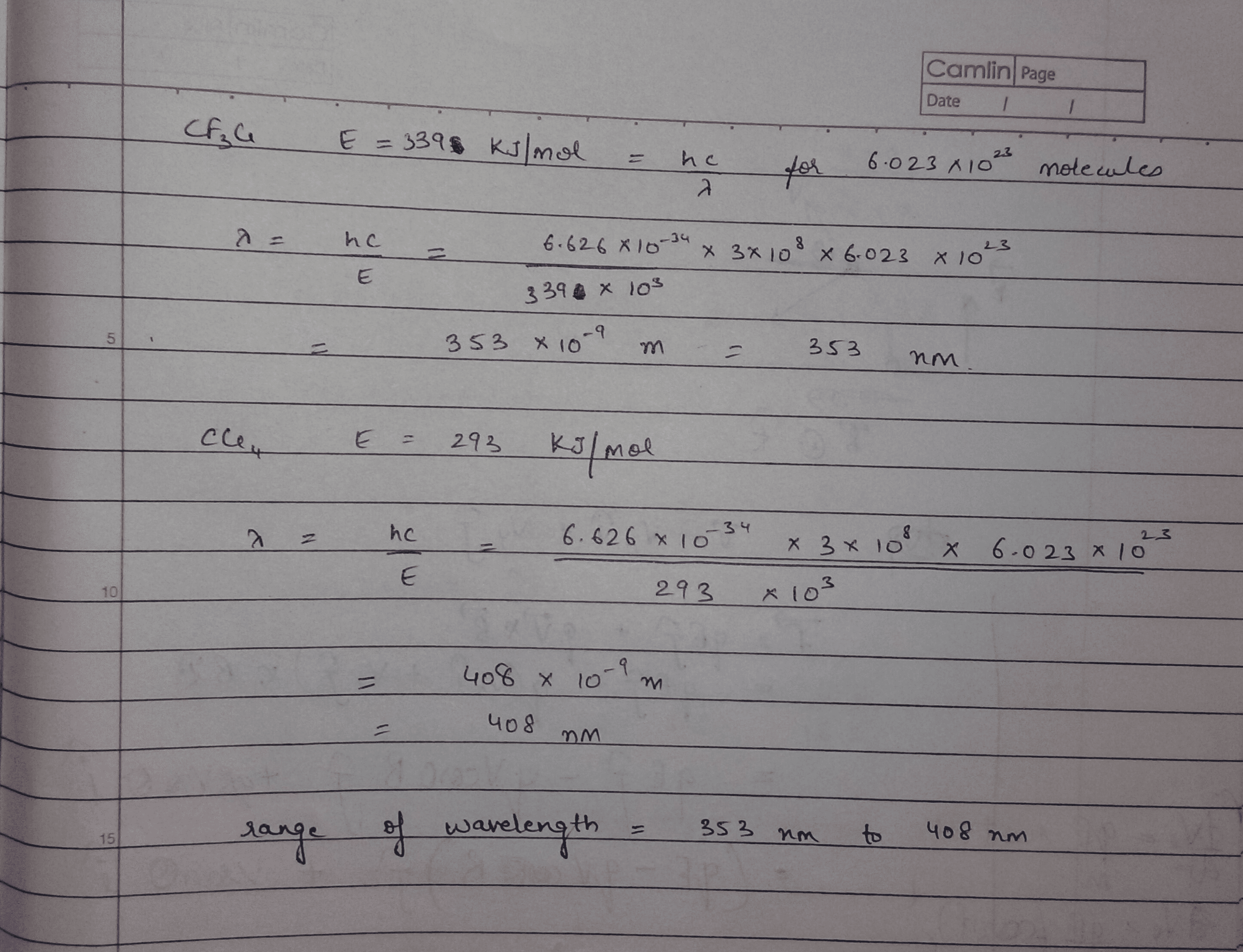
Oneclass In Cf3cl The Caˆ Cl Bond Dissociation Energy Is 339 Kj Mol In Ccl4 The Ccl Bond Dissociat

Schematic Potential Energy Diagram Illustrating Da To Cf 2 Cl 2 Download Scientific Diagram

Color Online Potential Energy Diagram For The Electron Capture Process Download Scientific Diagram

Photoionization Yield Curves For The Four Polyatomic Molecular Ions Download Scientific Diagram

Calculate C Cl Bond Enthalpy From Following Reaction Ch3cl G Cl2 G Ch2cl2 G Hcl G Dh O 104kj If C H Cl
Does Bond Length Follows This Order F2 Cl2 Br2 I2 My Professor Says The Correct Order Is F2 I2 Br2 Cl2 Quora
Comments
Post a Comment